Keep your kids safe when seconds matter
Learn life saving first aid from an experienced paramedic. Join over fifty thousand Australian parents and caregivers.
 Trusted across Australia
Trusted across Australia
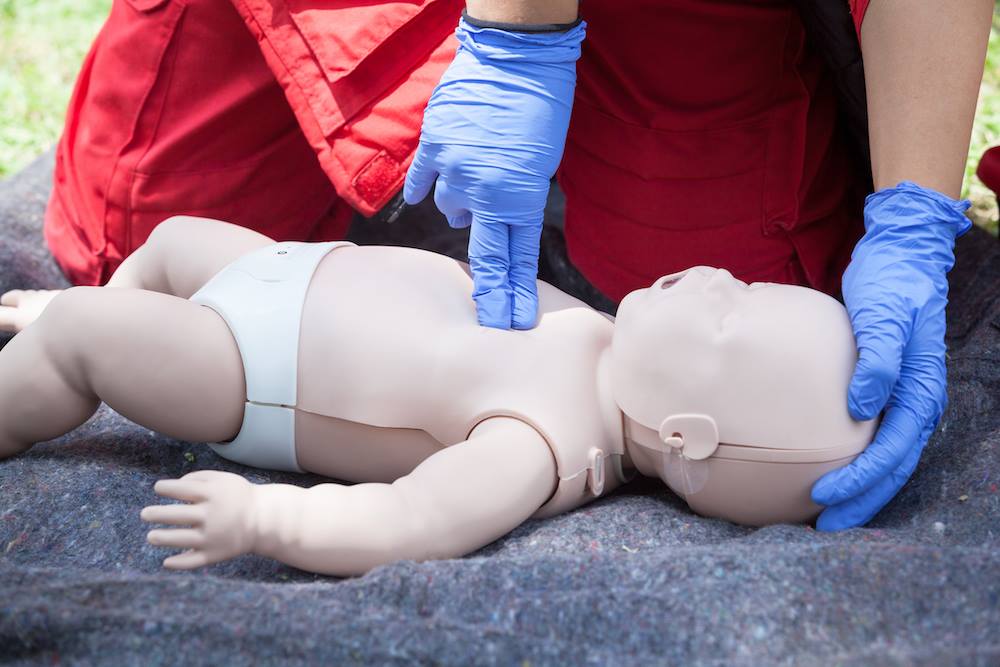

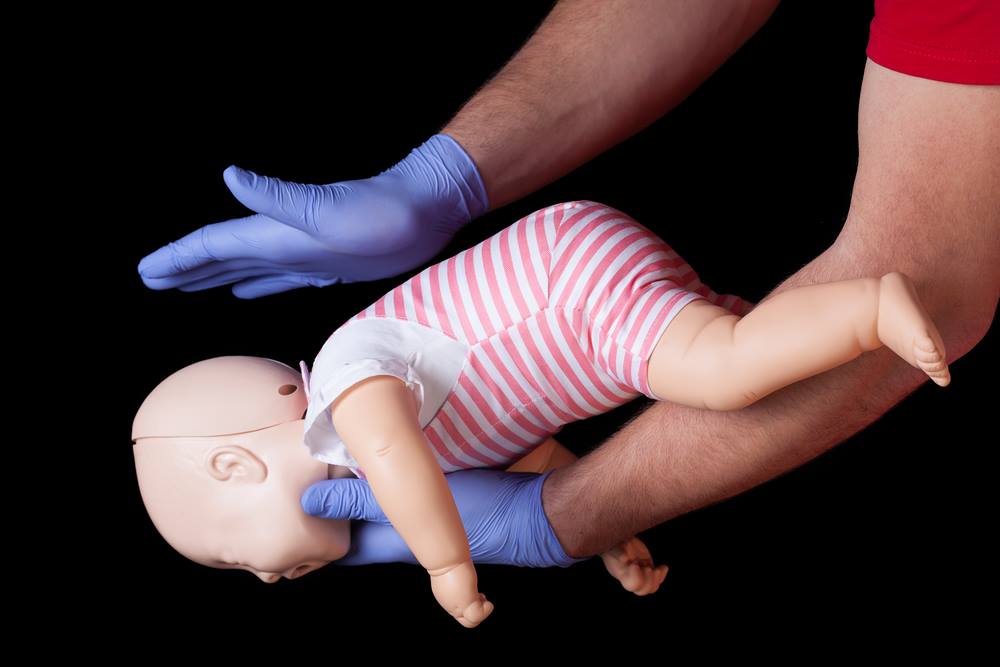
Why this matters
Every year, one in thirteen Australian children is hospitalised not from illness but from preventable injuries at home.
That number is higher than cases of cancer, asthma or infectious diseases combined. This course gives parents and caregivers the knowledge to act with calm and confidence, turning panic into protection.
- Simple steps for the first minute of any emergency
- Short lessons you can watch again anytime
- Real advice from a working paramedic
Meet Ben Fisher
A frontline paramedic with more than twenty years of real experience who has taught over fifty thousand parents across Australia.
You cannot rehearse an emergency but you can be ready for one
Ben created this course to make lifesaving knowledge simple calm and accessible. He has seen how panic steals time and how confidence gives it back.
 Trusted by families across Australia
Trusted by families across Australia


Inside the course
Short videos. Clear steps. Skills you remember when it counts.
- 1DRSABCD for any emergency
- 2Infant and child CPR
- 3Choking relief
- 4Drowning response
- 5Allergies and anaphylaxis
- 6Fits and fevers
- 7Meningococcal warning signs
- 8Breathing difficulties and asthma
- 9Burns and scalds
- 10Trauma and concussion
- 11Poisoning
- 12Bites and stings
- Instant email access after checkout
- Watch on any device at any time
- Course completion credential
- Trusted by more than fifty thousand families
Questions parents ask
Simple answers from a first response paramedic team. If you need anything else, we will look after you.
